 Conversely, for a given alkali metal ion, the fluoride salt always has. While in the case of NaF and KCl, NaF has a greater lattice energy compared to KCl because the size of the cation and anion is smaller in the case of KCl. Because the ionic radii of the cations decrease in the order K+ > Na+ > Li+ for a given halide ion, the lattice energy decreases smoothly from Li+ to K+. Among the given substances, MgO has the highest lattice energy compared to CaO as lattice energy is directly related to the charge and size of the cation. LiF, LiCl Which compound has higher lattice energy: BeS or. Figure 4.2.2 A Plot of Lattice Energy versus the Identity of the Halide for the Lithium, Sodium, and Potassium Halides. Which compound in each of the following pairs of ionic substances has the most exothermic lattice energy Justify your answers. Press 'calculate' to work out the different ionic. Lattice enthalpy of NaCl is 787 KJ/mol Lattice enthalpy of NaOH is 898 KJ/ mol. 
Choose the cation, anion and structure type from the lists provided or choose your own values for the ion charges and radii, the structure type and the value of n. Which compound has higher lattice energy: BaBr2 or CaBr2 Explain. This calculator should be used in conjunction with the notes on Understanding Crystal Structures. Generally, lattice energy is inversely proportional to the size of the ions and. Hence, it is typically used to measure the bond strength of ionic compounds. That means that we will have to use theoretical values of their lattice enthalpies. Which compound has the higher lattice energy: BaBr2 or CaBr2 Explain. Lattice energy can be defined as a measure of the energy required to dissociate one (1) mole of an ionic compound into its constituent anions and cations, in the gaseous state. Let's look at this in terms of Born-Haber cycles of and contrast the enthalpy change of formation for the imaginary compounds MgCl and MgCl 3. It turns out that MgCl 2 is the formula of the compound which has the most negative enthalpy change of formation - in other words, it is the most stable one relative to the elements magnesium and chlorine. Given Here, Enthalpy of sublimation of Potassium 89.0 kJ/mol Ionization Energy for Potassium IE (K) 419 kJ/mol Electron affinity for Chlorine is EA (Cl) 349 kJ/mol Bond energy of Chlorine, BE (Cl) 243 kJ/mol Enthalpy of formation for KCl, 436.5 kj/mol. The question arises as to why, from an energetics point of view, magnesium chloride is MgCl 2 rather than MgCl or MgCl 3 (or any other formula you might like to choose). The Lattice energy of KCl is -717 kJ/mol. (iii) The lattice energy affects the solubility of the ionic compounds.\( \newcommand\) (ii) The lattice energy is greater for small, highly charged ions. 
(i) The greater the lattice enthalpy, the more the stability of ionic compounds. 719 rank the following compounds by lattice energy: KF, KBr, KI, KCl. The important consequence of the lattice energies are: use the data in the table below to calculate the lattice energy of KCl. 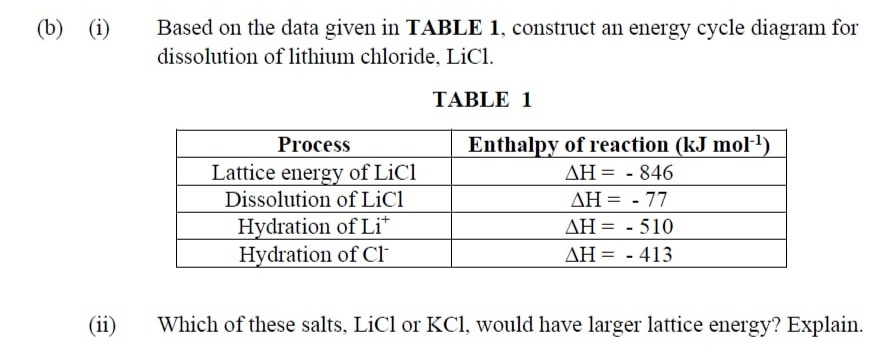
$\therefore $The order of lattice energy is $NaF > NaCl > KCl > RbCl$. The given ionic solids are $NaCl,$ion so $NaCl$has greater lattice energy than $KCl$ and $RbCl$. Hence, more will be the value of lattice energy. 
smaller the size of ions, lesser is the internuclear distance and greater will be the interionic attraction. larger the magnitude of charge on ions, greater will be the attractive forces, higher is the value of lattice energy and inversely proportional to size of an ion i.e. Lattice energy is directly proportional to charge on ion i.e. Lattice energy depends upon charge and size of cation. Hint: Lattice energy: The amount of energy released when $1$mole of an ionic solid is formed from its gaseous ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
